In-Use & Safety In-Use Testing
MD Supervised In-Use Studies
Dermatologist Supervised Safety In-Use Testing
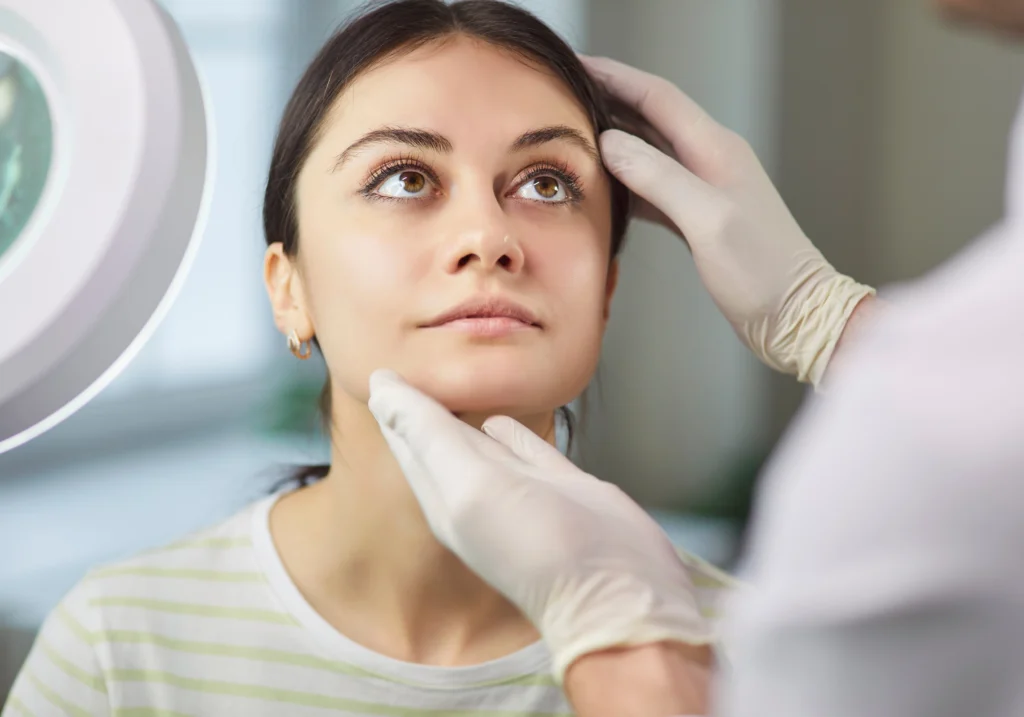

Study Characteristics
- Typically 20–50 subjects (customizable)
- Typical duration of 14–28 days (adaptable to product type)
- Used at-home under normal, real-life conditions
- First application supervised on-site to ensure correct use
- Self-assessment questionnaire-based evaluation
- Supervised by a certified medical professional, in this case a Dermatologist
- Monitored and evaluated for adverse reactions and tolerance
- Report delivery in 5 business days (express) or 10 (standard)

Typical Claim Support Areas
- Dermatologically tested/ supervised
- Clinically tested under Dermatologist supervision
- Dermatologist signed-off
- Perceived efficacy or visible results
- Ease of application and comfort after use
- Overall satisfaction and product acceptance
Ophthalmologist Supervised Safety In-Use Testing
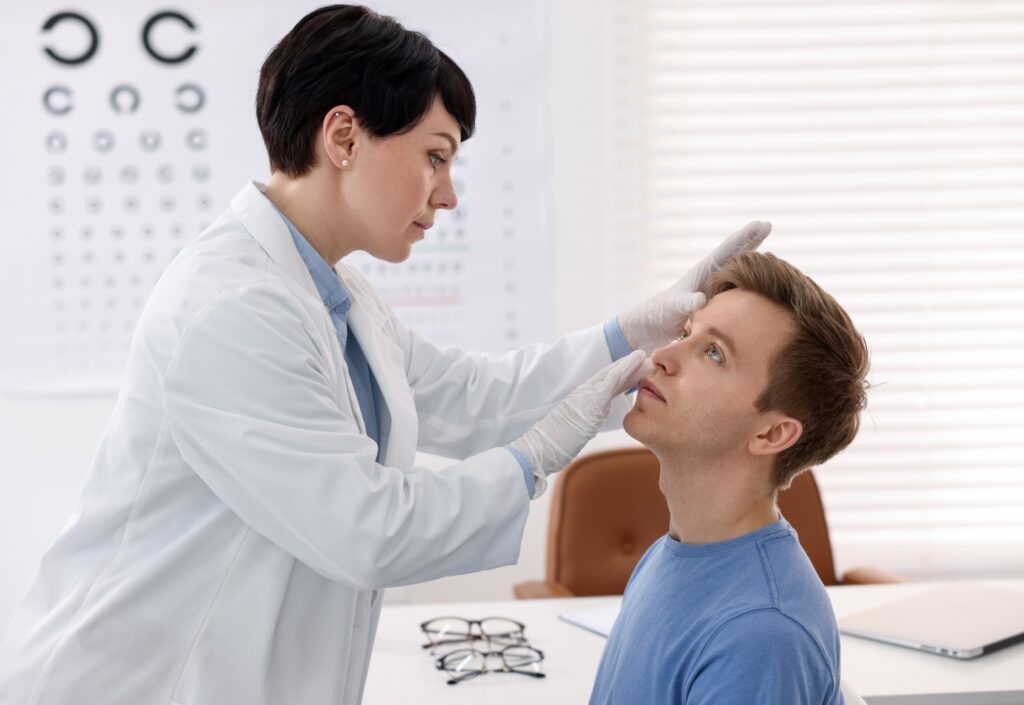

Study Characteristics
- Typically 20–50 subjects (customizable)
- Typical duration of 14–28 days (adaptable to product type)
- Used at-home under normal, real-life conditions
- First application supervised on-site to ensure correct use
- Self-assessment questionnaire-based evaluation
- Supervised by a certified medical professional, in this case an Ophthalmologist
- Monitored and evaluated for adverse reactions and tolerance
- Report delivery in 5 business days (express) or 10 (standard)

Typical Claim Support Areas
- Ophthalmologically tested/supervised
- Improves skin elasticity and/or firmness
- Clinically tested under ophthalmologist supervision
- Ophthalmologist signed-off
- Perceived efficacy or visible results around eye area
- Ease of application and comfort after use
- Overall satisfaction and product acceptance
Gynecologist Supervised Safety In-Use Testing


Study Characteristics
- Typically 20–50 subjects (customizable)
- Typical duration of 28 days (adaptable to product type)
- Used at-home under normal, real-life conditions
- First application supervised on-site to ensure correct use
- Self-assessment questionnaire-based evaluation)
- Supervised by a certified medical professional, in this case a Gynecologist
- Monitored and evaluated for adverse reactions and tolerance
- Report delivery in 5 business days (express) or 10 (standard)

Typical Claim Support Areas
- Gynecologist tested/supervised
- Clinically tested under gynecologist supervision
- Gynecologist tested for intimate use
- Perceived efficacy or comfort in intimate areas
- Safe for daily use in intimate zone
Odontologist Supervised Safety In-Use Testing
Our odontologist-supervised safety in-use studies evaluate how your oral care product is tolerated during realistic daily use. Board-certified odontologists combine clinical assessments with consumer feedback to validate safety claims with medical authority. Ideal for toothpastes, mouthwashes, lip balms, and peri-oral skincare, these studies deliver both regulatory compliance and consumer trust through professional oversight.
Most odontological clinical studies are conducted at specialized dental clinics due to the need for specialized equipment. The studies are carried under the odontologist upervision and are monitored by AQURA Labs staff.
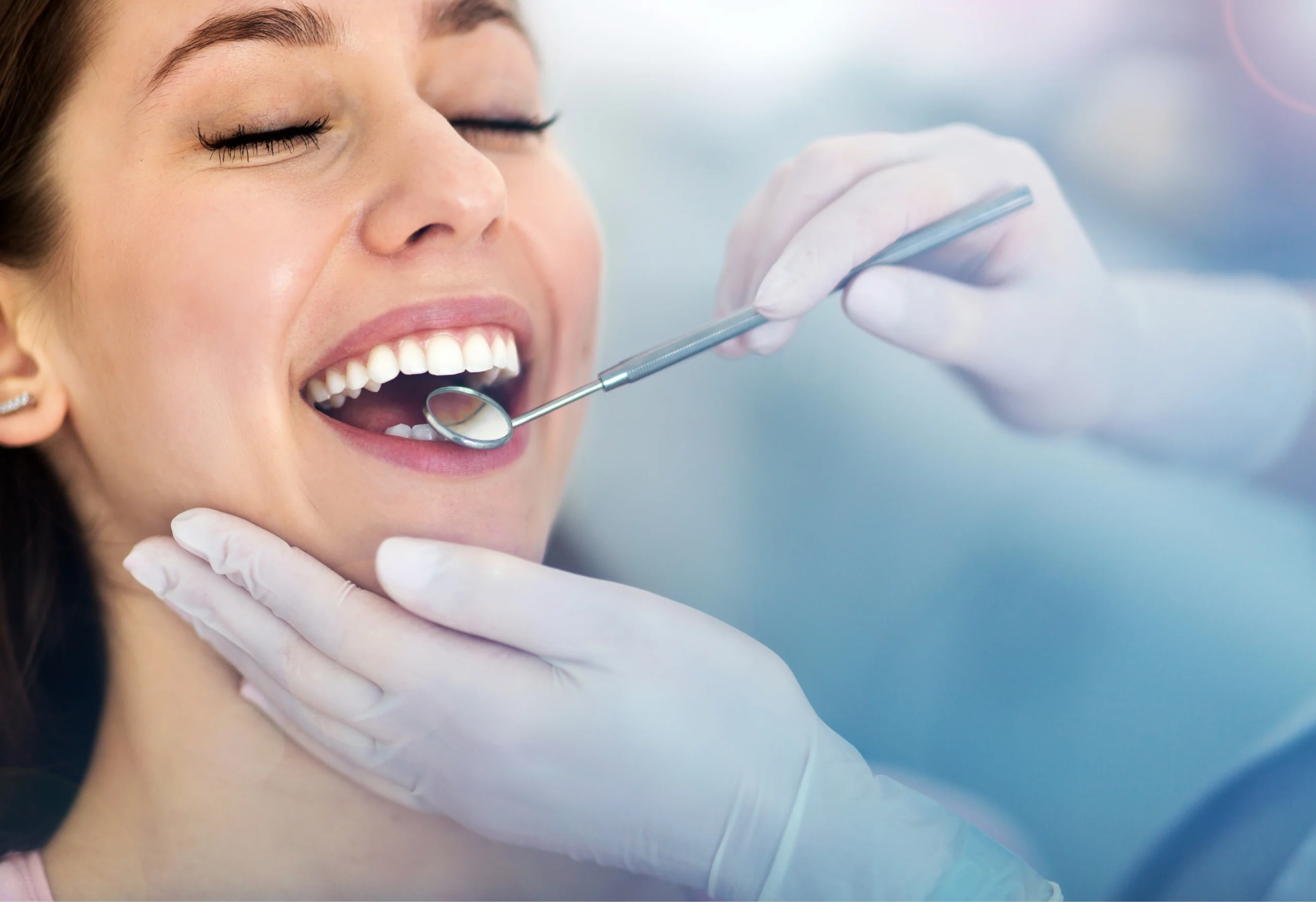

Study Characteristics
- Typically 20–50 subjects (customizable)
- Can be tested head to head (with other product) or vs. placebo
- Typical duration of 28 days (adaptable to product type)
- Used at-home under normal, real-life conditions
- First application supervised on-site to ensure correct use
- Self-assessment questionnaire-based evaluation)
- Supervised by a certified medical professional, in this case an Odontologist
- Monitored and evaluated for adverse reactions and tolerance
- Report delivery in 5 business days (express) or 10 (standard)

Typical Claim Support Areas
- Odontologist tested/supervised
- Clinically tested under odontologist supervision
- Odontologist tested for oral use
- Odontologist approved for daily oral care
- Perceived efficacy or comfort in oral cavity
- Safe for daily use in mouth/peri-oral zone
Multi-Specialist Clinical Supervision
Clinical studies don’t have to be single-specialty – we customize multi-MD supervision to match your product’s complexity and claims. Board-certified specialists collaborate from protocol design through final sign-off, delivering comprehensive safety data that regulators love.
Complex products touch multiple body areas or populations. One expert covers one domain; combined expertise covers all risks.
You can take advantage of conducting a single clinical study supervised by two medical doctors, thereby optimizing the study.

Examples
Eye Contour Cream (Dermatologist and Ophthalmologist supervised)
Dermatologist assesses peri-orbital skin tolerance.
Ophthalmologist evaluates eye safety (migrating product, tear film impact).
Claim example: “Safe for eye contour – Dermatologist and Ophthalmologist supervised”
Baby Lotion (Pediatrician and Dermatologist supervised)
Derm assesses any potential skin reactions
Pediatrician monitors infant-specific reactions (diaper zone, face)
Claim example: “Safe for babies – Pediatrician + Dermatologist verified”
Intimate + Perineal Postpartum Cream (Gynecologist and Dermatologist supervised)
Gynecologist evaluates mucous membrane tolerance
Dermatologist assesses external perineal skin
Claim example: “Postpartum safe for intimate use” – Gynecologist and Dermatologist supervised“
Dermatologist Supervised Safety In-Use Testing


Study Characteristics
- Typically 20–50 subjects (customizable)
- Typical duration of 14–28 days (adaptable to product type)
- Used at-home under normal, real-life conditions
- First application supervised on-site to ensure correct use
- Self-assessment questionnaire-based evaluation
- Supervised by a certified medical professional, in this case a Dermatologist
- Monitored and evaluated for adverse reactions and tolerance
- Report delivery in 5 business days (express) or 10 (standard)

Typical Claim Support Areas
- Dermatologically tested/ supervised
- Clinically tested under Dermatologist supervision
- Dermatologist signed-off
- Perceived efficacy or visible results
- Ease of application and comfort after use
- Overall satisfaction and product acceptance
Ophthalmologist Supervised Safety In-Use Testing


Study Characteristics
- Typically 20–50 subjects (customizable)
- Typical duration of 14–28 days (adaptable to product type)
- Used at-home under normal, real-life conditions
- First application supervised on-site to ensure correct use
- Self-assessment questionnaire-based evaluation
- Supervised by a certified medical professional, in this case an Ophthalmologist
- Monitored and evaluated for adverse reactions and tolerance
- Report delivery in 5 business days (express) or 10 (standard)

Typical Claim Support Areas
- Ophthalmologically tested/supervised
- Improves skin elasticity and/or firmness
- Clinically tested under ophthalmologist supervision
- Ophthalmologist signed-off
- Perceived efficacy or visible results around eye area
- Ease of application and comfort after use
- Overall satisfaction and product acceptance
Pediatrician Supervised Safety In-Use Testing
Our pediatrician-supervised safety in-use studies evaluate how your children’s product is tolerated during realistic daily use. Board-certified pediatricians combine clinical assessments with parent feedback to validate safety claims with medical authority. Ideal for baby lotions, diaper creams, and children’s skincare, these studies deliver both regulatory compliance and consumer trust through professional oversight.
Prior to testing on children additional safety testing (e.g., on adults with sensitive skin) may be required.


Study Characteristics
- Typically 20–50 subjects (customizable)
- Typical duration of 28 days (adaptable to product type)
- Can be tested on different age groups ( eg. infants, toddlers etc.)
- Used at-home under normal, real-life conditions
- First application supervised on-site to ensure correct use
- Assessment questionnaire-based evaluation (typically filled-out by the parents
- Supervised by a certified medical professional, in this case a Pediatrician
- Monitored and evaluated for adverse reactions and tolerance
- Report delivery in 5 business days (express) or 10 (standard)

Typical Claim Support Areas
- Pediatrician tested/supervised
- Clinically tested under pediatrician supervision
- Pediatrician sign-offed
- Perceived efficacy or visible results on children’s skin
- Overall satisfaction and product acceptance
Gynecologist Supervised Safety In-Use Testing


Study Characteristics
- Typically 20–50 subjects (customizable)
- Typical duration of 28 days (adaptable to product type)
- Used at-home under normal, real-life conditions
- First application supervised on-site to ensure correct use
- Self-assessment questionnaire-based evaluation)
- Supervised by a certified medical professional, in this case a Gynecologist
- Monitored and evaluated for adverse reactions and tolerance
- Report delivery in 5 business days (express) or 10 (standard)

Typical Claim Support Areas
- Gynecologist tested/supervised
- Clinically tested under gynecologist supervision
- Gynecologist tested for intimate use
- Perceived efficacy or comfort in intimate areas
- Safe for daily use in intimate zone
Odontologist Supervised Safety In-Use Testing
Our odontologist-supervised safety in-use studies evaluate how your oral care product is tolerated during realistic daily use. Board-certified odontologists combine clinical assessments with consumer feedback to validate safety claims with medical authority. Ideal for toothpastes, mouthwashes, lip balms, and peri-oral skincare, these studies deliver both regulatory compliance and consumer trust through professional oversight.
Most odontological clinical studies are conducted at specialized dental clinics due to the need for specialized equipment. The studies are carried under the odontologist upervision and are monitored by AQURA Labs staff.


Study Characteristics
- Typically 20–50 subjects (customizable)
- Can be tested head to head (with other product) or vs. placebo
- Typical duration of 28 days (adaptable to product type)
- Used at-home under normal, real-life conditions
- First application supervised on-site to ensure correct use
- Self-assessment questionnaire-based evaluation)
- Supervised by a certified medical professional, in this case an Odontologist
- Monitored and evaluated for adverse reactions and tolerance
- Report delivery in 5 business days (express) or 10 (standard)

Typical Claim Support Areas
- Odontologist tested/supervised
- Clinically tested under odontologist supervision
- Odontologist tested for oral use
- Odontologist approved for daily oral care
- Perceived efficacy or comfort in oral cavity
- Safe for daily use in mouth/peri-oral zone
Multi-Specialist Clinical Supervision
Clinical studies don’t have to be single-specialty – we customize multi-MD supervision to match your product’s complexity and claims. Board-certified specialists collaborate from protocol design through final sign-off, delivering comprehensive safety data that regulators love.
Complex products touch multiple body areas or populations. One expert covers one domain; combined expertise covers all risks.
You can take advantage of conducting a single clinical study supervised by two medical doctors, thereby optimizing the study.

Examples
Eye Contour Cream (Dermatologist and Ophthalmologist supervised)
Dermatologist assesses peri-orbital skin tolerance.
Ophthalmologist evaluates eye safety (migrating product, tear film impact).
Claim example: “Safe for eye contour – Dermatologist and Ophthalmologist supervised”
Baby Lotion (Pediatrician and Dermatologist supervised)
Derm assesses any potential skin reactions
Pediatrician monitors infant-specific reactions (diaper zone, face)
Claim example: “Safe for babies – Pediatrician + Dermatologist verified”
Intimate + Perineal Postpartum Cream (Gynecologist and Dermatologist supervised)
Gynecologist evaluates mucous membrane tolerance
Dermatologist assesses external perineal skin
Claim example: “Postpartum safe for intimate use” – Gynecologist and Dermatologist supervised“
Our in-use studies evaluate how your product performs and is tolerated under typical daily use. By combining consumer feedback with medical oversight, they help validate both subjective efficacy and safety-related claims. Particularly relevant for leave-on products or sensitive areas, these studies bridge real-world experience with professional assessment.


Study Characteristics
- Typically 20–50 subjects (customizable)
- Typical duration of 14–28 days (adaptable to product type)
- Used at-home under normal, real-life conditions
- First application supervised on-site to ensure correct use
- Self-assessment questionnaire-based evaluation
- Supervised by a certified medical professional, such as a dermatologist or ophthalmologist
- Monitored and evaluated for adverse reactions and tolerance
- Report delivery in 5 business days (express) or 10 (standard)

Typical Claim Support Areas
- Dermatologically and/or ophthalmologically tested
- Gentle formula suitable for daily use
- Feel or comfort during or after application
- Perceived efficacy or visible results
- Ease of application and comfort after use
- Overall satisfaction and product acceptance