Regulators ask for “safety substantiation,” but what actually matters most in practice are the studies you run on real people. This article walks step by step through subject-based safety testing, from basic patch tests to robust safety‑in‑use studies, with a pragmatic EU–US perspective.
First Line of Defense: Patch Testing
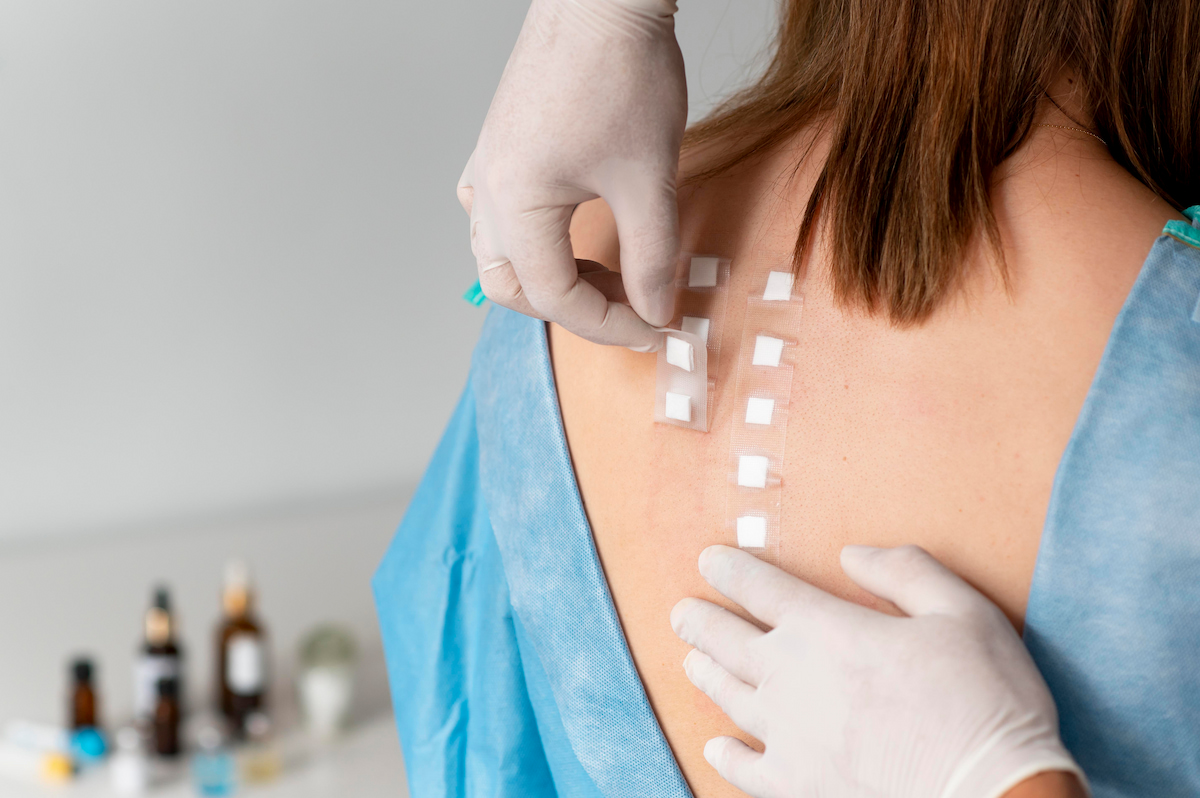
For most topical cosmetics, the first subject study is a patch test to screen for irritation (and sometimes sensitization) under controlled, exaggerated conditions.
- In the EU, it’s common practice to run a simple occlusive or semi-occlusive patch test in a small panel, ranging from 10–30 healthy adults with a 24-hour occlusion, though it is recommended to include at least 20–30 subjects to confirm that a finished cosmetic does not cause unacceptable acute irritation on intact skin.
- For low‑risk, well‑characterized formulas, this kind of basic patch test, combined with a solid safety assessment and supporting toxicology data, is often considered sufficient as an initial human safety check.
In the US, brands and labs often go a step further and use HRIPT (which is a type of Patch Test – Human Repeat Insult Patch Test) on larger panels (commonly 50+ subjects) under repeated occlusion.
- HRIPT is widely treated as the “gold standard” for in‑vivo irritation/sensitization screening and is frequently requested by major retailers (e.g. Amazon or Walmart).
- It also helps assess allergenicity potential, because any sensitization that occurred during induction will show up as a reaction at the challenge site after the rest/memory phase. Used alongside a thorough ingredient safety review, HRIPT data can help you support the “hypoallergenic” claim.
- Importantly, current practice indicates that HRIPT is not explicitly mandated by law for cosmetics; it is a strong market practice and risk‑management tool, rather than a test specified line‑by‑line in regulation.
For low-risk cosmetics in the EU, a well-conducted 20–30 subject patch test is often a sufficient first human step. In the US, HRIPT isn’t legally required but is the market practice “gold standard” widely used to signal strong skin-safety standards, especially to major retailers.
Going Beyond Patches: Why Safety-in-Use Studies Matter
Patch tests answer a narrow question: “Does this product irritate or sensitize skin under exaggerated, controlled conditions?” To show that a product is truly safe in real life, you need safety‑in‑use (SIU) studies that mirror how consumers actually use the product and apply it under “normal conditions of use”.
Best practice is to:
- Run SIU with study subject under realistic or “normal conditions of use” according to instructions (or at a modestly higher frequency). The study should be run for 28 days (a month of product use) and always on the intended body site.
- Include both normal and self‑perceived sensitive skin where relevant, to better reflect real‑world users. If the product is destined for a population with sensitive skin, you can recruit a study panel with 100% self-perceived sensitive skin.
- Capture both objective and subjective safety
- Objective assessments (erythema, dryness, scaling, visible irritation) performed by a trained clinician.
- Subject‑reported sensations (stinging, burning, tightness, discomfort), collected via structured questionnaires.
For safety‑in‑use studies, a duration of about 28 days is often used so that any cumulative effects or delayed reactions can emerge under real‑world conditions. For many regulators, retailers and brand owners, this type of study is what closes the loop between theoretical safety and practical safety: “Does it behave well when people actually use it?”

Medical Oversight: Dermatologists and Beyond
The credibility of a safety study depends not just on the protocol, but on who is supervising it.
Dermatologist supervision as the baseline
For skin‑applied cosmetics, Dermatologist‑supervised safety‑in‑use studies are now close to a must‑have if you want robust, challenge‑proof data.
In a typical study design:
- A dermatologist performs clinical skin tolerance assessments at baseline and at the final visit, scoring redness, dryness, scaling, edema or other reactions on standardized scales.
- The dermatologist also reviews any adverse events reported between visits and determines if they constitute adverse reactions related to the product.
This level of oversight supports both safety substantiation and carefully worded “dermatologist tested” or “dermatologist supervised” claims.
Specialists for specific body areas
- Ophthalmologist – eye area products. For eye creams, mascaras, eyeliners, and makeup removers, an ophthalmologist can assess eyelids, conjunctiva, and cornea, ensuring the product is well tolerated around and inside the peri-ocular area.
- Gynecologist – intimate hygiene products. For vulvar or vaginal hygiene products, lubricants, or related applications, a gynecologist can evaluate local mucosal tolerance, pH compatibility, and any impact on comfort or the intimate area.
- Pediatrician – products for children. For products specifically intended for babies and children (wipes, creams, shampoos or even nappies), a pediatrician can help judge age‑appropriate use, dosing frequency, and the relevance of any observed reactions in a younger population.
- Odontologist – oral care and perioral products. For toothpaste, mouthwash, whitening treatments, or products used around the lips and gums, an odontologist (dental specialist) can evaluate gingival and oral‑mucosa tolerance, tooth surface effects, and any sensitivity; many of these studies are best run in a dental clinic, where the specialized equipment and diagnostic tools are already in place.
When you combine experts, for example a Dermatologist and Ophthalmologist supervised safety‑in‑use study, for an eye cream (applied in the peri-ocular area) you get a more robust evaluation, with each physician assessing safety from their own specialty perspective. These multi‑specialist designs allow you to maximize the study design, efficiently using participants and resources, and generating rich, multi‑angle safety data that underpins stronger and more defensible claims.

Putting it Together: A Stepwise Human Safety Program
For most new cosmetic launches, a pragmatic, defensible human safety program can be built in three steps:
- Initial patch test (EU and US)
- EU: start with a basic singe application patch test on 10–30 healthy adults (with 20-30 recommended) to screen for acute irritation under occlusion or semi‑occlusion.
- US: consider HRIPT on a larger panel if you want to align with common US market practice.
- Dermatologist‑supervised safety‑in‑use study
- Run a SIU study under realistic use on the intended site, with clinical skin tolerance assessments performed by the Dermatologist and a self-assessment performed by the study subjects.
- Include sensitive‑skin subjects where appropriate and ensure follow‑up is long enough (often 28 days) to capture cumulative effects.
- Specialist‑enhanced SIU where needed
- If you are testing a specific product and an evaluation by multiple MDs is possible, it is recommended to do so as it strengthens the robustness of the study.
The best practice especially for products targeting sensitive skin, the eye area, intimate hygiene, children, or oral use is a robust safety‑in‑use program, ideally supervised by a Dermatologist and one or more relevant specialists. These types of studies let you optimize your design and visits while delivering very strong, multi‑angle safety outcomes.
Why This Level of Safety Assessment Matters
All of this work has a very simple business goal: introduce safe, well‑tolerated products on the market and minimize future risks. A single severe skin or eye reaction, especially if it circulates on social media, can damage a brand’s reputation far more than the cost of a robust safety testing.
Safety‑in‑use studies, especially when supervised by a Dermatologist and, where appropriate, a second MD (such as an Ophthalmologist, Gynecologist, Pediatrician, Odontologist etc.), provide the best multi‑angle view of safety you can realistically achieve. They combine clinical observation with subject experience and site‑specific expertise, giving you a safety story that protects consumers, supports your regulatory obligations and safeguards the long‑term equity of your brand.